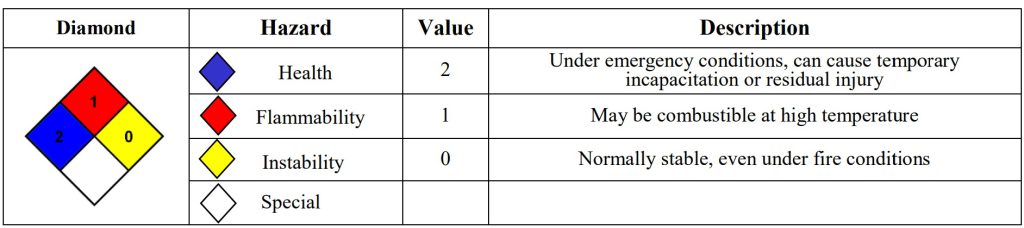
Physical properties
Physical state: liquid
Color: colorless
Melting point: -13 °C (8.6°F)
Density: 1.115 g/cm3; 20°C
Chemical properties
Molecular formula: C2H6O2
Molecular mass: 62.068 g/mol
Boiling point: 197.3°C (387.1°F )
Solubility in water:
20°C; completely miscible
| Flammability limit | Threshold limit value | ||
|---|---|---|---|
| LFL | UFL | TLV-TWA | TLV-STEL |
| 3.2% | 15.3% | 25 ppm | 50 ppm. 10mg/m3 |
Mono ethylene glycol: General description And Use
USE
- Used to make antifreeze and de-icing solutions for cars, airplanes, and boats.
- Precursor to polymers.
- Dehydrating agent in the natural gas industry.
- Hydrate formation inhibitor in the gas industry.
- Manufacturing of capacitors.
- Solvent.
- Used in hydraulic brake fluids and inks used in stamp pads, ballpoint pens, and print shops.
- Manufacturing of polyester fibers.
General description
Mono ethylene glycol (also known as MEG, EG, 1,2-ethanediol or, 1,2 Dihydroxyethane) is an organic compound with the formula C2H6O2. It is a slightly syrupy viscous liquid with a clear, colorless appearance, and a sweet taste (but it is toxic in high concentrations) that emits virtually no odor and absorbs water easily. It’s miscible with water, alcohols, and many other organic compounds. The primary hazard is its threat to the environment; since it is a liquid that can easily penetrate the soil and contaminate groundwater and nearby streams. To prevent this, immediate steps should be taken to limit its spread.
Mono ethylene glycol: Warnings and caution
GHS hazard statements
H373 – May cause damage to organs (kidneys) through prolonged or repeated exposure.
H302– Harmful if swallowed
GHS precautionary statements
P260 – Do not breathe dust/fume/gas/mist/vapor/spray.
P264 – Wash hands, forearms, and face thoroughly after handling.
P270 – Do not eat, drink or smoke when using this product.
P301 + P312- IF SWALLOWED: Call a POISON CENTER or a physician if you feel unwell.
P330 -Rinse mouth.
P501– Dispose of containers to an approved waste disposal plant.